On World Children’s Day, WHO announced an issue of a call urging stakeholders to accelerate access to effective paediatric HIV and tuberculosis (TB) diagnostics and medicines.
The Action Plan, which was launched on the 20th of November 2020, has been developed by a wide group of stakeholders under the auspices of the Fifth V*****n High-Level Dialogue on Paediatric HIV and TB in Children Living with HIV which was held earlier this month.
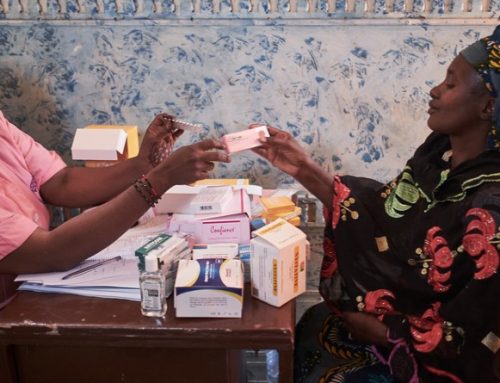
WHO reported that Children are one of the most disadvantaged populations in the HIV and AIDS and TB response. In 2019, 95 000 AIDS-related deaths occurred in children, two-thirds of those deaths in 21 focus countries. 850 000 children living with HIV were not accessing treatment, 65% of which were aged 5-14 years. These children are also particularly susceptible to co-infection with tuberculosis, a major cause of AIDS-related deaths in this population. In 2019, an estimated 36 000 children who were living with HIV died from TB.
There are several challenges that hamper the rapid development of paediatric formulations, including lack of paediatric data for new drugs, delay in completion of clinical studies, challenges with taste, and slow market uptake among others.
The plan agreed upon by participants of the High Level Dialogue includes pledges to accelerate development of new pediatric HIV and TB formulations; improved diagnostic devices and assays for children with TB; and lower prices for early infant HIV diagnosis.
“The impact of the COVID-19 pandemic has l**d bare the power of collaboration and partnership to accelerate action. The WHO Global HIV programme recognizes this Action Plan as the roadmap to reset the speed at which innovations in drugs and diagnostics can lead to child-centered impact. We are proud to commit to developing the norms and standards, policies and research agendas on this pathway to success” said Dr Meg Doherty, Director the WHO Global HIV, Hepatitis and STI Programmes.
For more information, check the full article at: https://www.who.int/news/item/20-11-2020-accelerate-access-to-effective-paediatric-hiv-and-tuberculosis-diagnostics-and-medicines






















































Leave A Comment
You must be logged in to post a comment.